A) 47.8%
B) 58.4%
C) 32.4%
D) The identity of the compound must be known.
E) none of the above
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the solubility of sodium acetate (Molar mass = 82 g/mol) is 76 grams per 100 grams of water,which of the following solutions would be considered supersaturated?
A) 8.5 moles of sodium acetate dissolved in 1 L of water
B) 5.5 moles of sodium acetate dissolved in 500 mL of water
C) 1.8 moles of sodium acetate dissolved in 300 mL of water
D) 1.2 moles of sodium acetate dissolved in 200 mL of water
E) none of the above
G) B) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Which of the following compounds is a strong electrolyte?
A) C6H12O6
B) C7H14O5
C) C4H8O2
D) NaC2H3O2
E) all of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of KOH are contained in 750.mL of 5.00 M KOH solution?
A) 6.67 mol
B) 3.75 × ![]()
Mol
C) 56.1 mol
D) 3.75 mol
E) none of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 90.0 g sample of NaOH is dissolved in water and the solution is diluted to give a final volume of 3.00 liters.The molarity of the final solution is ________.
A) 0.500 M
B) 0.750 M
C) 1.00 M
D) 2.25 M
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What molarity should the stock solution be if you want to dilute 25.0 mL to 2.00 L and have the final concentration be 0.103 M?
A) 0.206 M
B) 8.24 M
C) 4.12 M
D) 0.243 M
E) none of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mass percent of an ammonium carbonate solution prepared by dissolving 33.2 grams of solid into 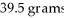 Of water?
Of water?
A) 84.1%
B) 72.7%
C) 45.7%
D) 54.3%
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Tap water contains dissolved nitrogen and oxygen.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
A stock solution is a more concentrated form than what is typically used in a lab.It often requires further dilution.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the ion concentrations in a 0.12 M solution of AlCl3?
A) 0.36 M Al3+ ions and 0.12 M Cl- ions
B) 0.12 M Al3+ ions and 0.040 M Cl- ions
C) 0.12 M Al3+ ions and 0.36 M Cl- ions
D) 0.040 M Al3+ ions and 0.040 M Cl- ions
E) none of the above
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of NaF are in 34.2 grams of a 45.5% by mass NaF solution?
A) 0.814
B) 75.2
C) 15.6
D) 0.371
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many mL of 0.112 M Pb(N O3) 2 are needed to completely react with 25.0 mL of 0.105 M KI? Given: Pb(NO3) 2 (aq) + 2KI(aq) → PbI2 (s) + 2KNO3(aq)
A) 11.7
B) 23.4
C) 0.147
D) 5.85
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A supersaturated solution is unstable and crystallization usually occurs.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The molarity of a solution prepared by dissolving 15.0 grams of NaCl in 1000 mL water is 0.15 M.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many grams of barium sulfate are produced if 25.34 mL of 0.113 M BaCl2 completely react given the reaction: BaCl2 (aq) + Na2SO4(aq) → BaSO4 (s) + 2NaCl (aq)
A) 5.90
B) 26.3
C) 1039
D) 0.668
E) none of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution contains 100.0 g water,10.0 g NaCl,and 15.0 g methanol.What is the weight percent of methanol in the solution?
A) 8.00%
B) 10.0%
C) 12.0%
D) 15.0%
E) none of the above
G) A) and E)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
When preparing sodium hydroxide solution,it is best to use water that does not contain any dissolved carbon dioxide as it reacts with the sodium hydroxide.Removing the carbon dioxide can be accomplished by:
A) vigorously stirring the solution.
B) using water fresh out of the purification system.
C) boiling the water.
D) Nothing can be done to remove dissolved gases.
E) none of the above
G) A) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Calculate the molarity of a KCl solution made by dissolving 24.7 g of KCl in a total volume of 500.mL.
A) 0.331
B) 0.663
C) 0.166
D) 6.04
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Osmosis is the process in which a solvent moves from an area of low solute concentration to an area of high solute concentration.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which solution below contains the highest total quantity of dissolved sodium ions?
A) 100.mL of 4.0 M NaCl
B) 75.0 mL of 3.0 M Na2SO4
C) 50.0 mL of 8.0 M NaOH
D) 50.0 mL of 2.0 M Na3PO4
E) none of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 110
Related Exams